The potential use of bacteria and bacterial derivatives as drug ... - Virology Journal
Novel approaches to treating viral illnesses must be developed. Drug delivery methods based on nanomaterials are attractive options in the search for therapeutic advantages because of their distinctive properties. Researchers should also examine how the most prevalent viruses, such as the flu, HIV, SARS-CoV-2, monkeypox, and HPVs, are being treated and prevented using the latest advances in nanomedicine [4, 17, 68, 69]. Bacterial entry into mammalian host cells involves various mechanisms, such as clathrin-mediated endocytosis, caveolin-mediated endocytosis, and membrane fusion. MVs, prominent spherical liposomes, play various roles in Gram-positive and Gram-negative bacteria. These roles include intercellular communication and the transportation of secreted proteins and virulence factors into the host or competing bacterial cells [70]. In addition, it is possible to utilize genetically modified bacteria to suppress particular infectious ailments [71].
The concept that the most effective approach to managing prevalent infectious diseases is by developing potent vaccines that do not require adjuvants has led to the exploration of diverse biological immune-stimulating constituents as novel vaccine contenders. In contemporary times, there has been a surge of interest in EVs, commonly referred to as exosomes and microvesicles in mammalian cells and OMVs in Gram-negative bacteria, as potential candidates for the next-generation vaccine. Nevertheless, the greater the invasiveness and efficacy of the vaccine in administration, the higher the likelihood of severe immune toxicity [72]. The immune system reacts aggressively to the live bacterial vector because of its inherently hostile characteristics. The mucosal method is used to deliver the live bacterial vector, which has the extra advantage of boosting the immune system both mucosally and systemically. The vaccination is straightforward to deliver through the mucosal route, which is less intrusive and increases patient acceptability. One of the most promising microorganisms utilized as a live bacterial vector is the lactic acid bacterium. Salmonella spp. and Shigella spp. are two examples of attenuated harmful bacteria employed as DNA vaccine carriers. Several studies have shown that live bacterial vectors are an effective method for delivering DNA vaccinations [73]. Furthermore, BG's potent tropism for APCs encourages the development of cellular and humoral responses to heterologous antigens and carrier- ENV complexes. BGs are well-suited for use as combination vaccines because of the ease with which they may be produced and packaged with (many) target antigens. Because they are freeze-dried, BG vaccines have a long shelf life and don't require cold-chain storage; they're risk-free because they don't use host DNA or live organisms; they're more effective at delivering their target antigens than traditional methods; they're flexible in terms of the DNA or protein antigens they can use; and they're highly bioavailable [56]. Even though BG have been studied as vaccine candidates for use against their own ENV structures, they are most often utilized as flexible carriers and adjuvant vehicles for antigens of bacterial or viral origin. A proficiently crafted bacterium engineered for drug delivery purposes comprises three fundamental constituents: the localization of the affected tissue and regulatory input cue, the intracellular genetic circuitry component, and the active compound delivery module [16]. The present study aimed to examine the potential of bacterial-mediated drug delivery systems in treating viral infections, including but not limited to hepatitis virus, coronavirus, HIV, influenza, and EV.
The utilization of bacteria as a delivery system in SARS-CoV-2
Coronaviruses (CoVs) are classified into four distinct phylogenetic groups, namely Alpha-, Beta-, Gamma-, and Delta-CoV. Additionally, human CoVs can be primarily categorized into two types, namely α and β-CoV [74,75,76]. SARS-CoV-2 is classified as a member of the β-coronavirus family and is responsible for the global COVID-19 pandemic [77, 78]. The virus is known to cause both upper and lower respiratory tract illnesses, including asymptomatic cases [79, 80]. CoVs are positive sense, single-stranded RNA (+SS-RNA) [81]. The viral genome contains multiple smaller open reading frames (ORFs). ORFs in question are predicted to be responsible for encoding various structural proteins, such as the spike (S) glycoprotein, ENV, membrane (M), nucleocapsid (N) proteins, as well as non-structural proteins (NSP) [82]. According to research, Salmonella strains have the potential to serve as a promising foundation for the creation of an oral vaccine for COVID-19. This approach could serve as a viable alternative for addressing the emergence of diverse mutated coronavirus strains and novel infectious diseases in the future. Bacteria-based vaccines can be developed through oral administration of bacterial strains that exhibit tolerance to the gastrointestinal tract. In a study, an attenuated strain of Salmonella (characterized by diminished virulence and toxicity) was investigated as a potential carrier for oral vaccines. The present strain was engineered with the aim of producing or partially releasing antigens against SARS-CoV-2 through the utilization of the sipB160 protein. Significantly, Salmonella has the potential to function as a vaccine by inducing activation of B- and T-cells through phagocytosis or viral infection of macrophages that are present in the intestinal region while maintaining favorable bacterial cell viability. The synthesis of three distinct antigens was conducted utilizing bioinformatics data about the genetic makeup of antigens to facilitate the development of COVID-19 vaccines. A plasmid using the lac operon was developed as a vector for expressing an antigen in Salmonella. Bacteria were transformed with the synthesized version of the intended plasmid. Here, the plasmid was first confirmed cloned in E. coli and then converted into a shuttle Salmonella strain and a vaccine-grade Salmonella strain. Three different coronavirus protein antigen candidates were generated to ensure the antigen was adequately expressed in the vaccination candidate strain [83].
OMV-based vaccinations will likely be much simpler to store and carry than, for example, mRNA vaccines; nonetheless, stability and effectiveness must be studied rigorously for each formulation. These qualities may advocate for the widespread use of OMV vaccines, particularly in regions where low-temperature refrigeration technology is scarce. Two other OMV-based SARS-CoV-2 vaccines have been brought to researcher attention since our preprint was published [84]. In a study, a novel SARS-CoV-2 vaccine candidate has been reported by researchers, which utilizes S. Typhimurium EVs decorated with the Spike receptor-binding domain (RBD) derived from mammalian cell culture. The Mesocricetus auratus, commonly known as the golden Syrian hamster, was utilized as a COVID-19 model and subsequently administered immunization via RBD-conjugated OMVs (RBD-OMVs). The administration of intranasal (i.n.) immunization elicited elevated levels of blood anti-RBD IgG and observable mucosal responses. The vaccinated individuals exhibited neutralizing antibody activity against wild-type and Delta variants. When exposed to live virus, hamsters that were immunized with RBD-OMV showed a significant reduction in body mass loss, lower virus titers in bronchoalveolar lavage fluid, and less severe lung pathology compared to animals that were immunized with unconjugated OMVs or vehicle control. The findings of this research underscore the significance and adaptability of vaccine strategies based on OMVs [85] (Fig. 4). In another study, an i.n. COVID-19 subunit vaccine was developed by Van Der Ley et al. based on a recombinant, six-proline-stabilized, D614G spike protein of SARS-CoV-2 connected through the LPS-binding peptide sequence mCramp (mC) to OMVs from Neisseria meningitidis (N. meningitidis). After the spike protein was produced in CHO cells and coupled to the OMVs, mice and Syrian hamsters received i.n. or intramuscular (i.m.) prime-boost immunizations with the OMV-mC-Spike vaccine. The administration of OMV-mC-Spike resulted in the production of serum-neutralizing antibodies in all vaccinated animals, as observed in this study. It is noteworthy that i.n. vaccination resulted in elevated levels of spike-binding IgG and IgA Abs in the nasal and pulmonary regions, whereas i.m. vaccination only elicited an IgG response in the serum. The study findings indicate that hamsters, who were administered the second vaccination dose against SARS-CoV-2, were safeguarded against weight loss and viral replication in the lungs. This was in contrast to the control groups that received OMV or spike alone. Hamsters vaccinated against OMV-mC-Spike exhibited no pathological lesions in the lungs 7 days after the challenge, in contrast to the unvaccinated controls. Positive results from clinical trials with the OMV-mC-Spike candidate vaccine indicate that this unique, needle-free, subunit vaccination idea should be developed further [86].
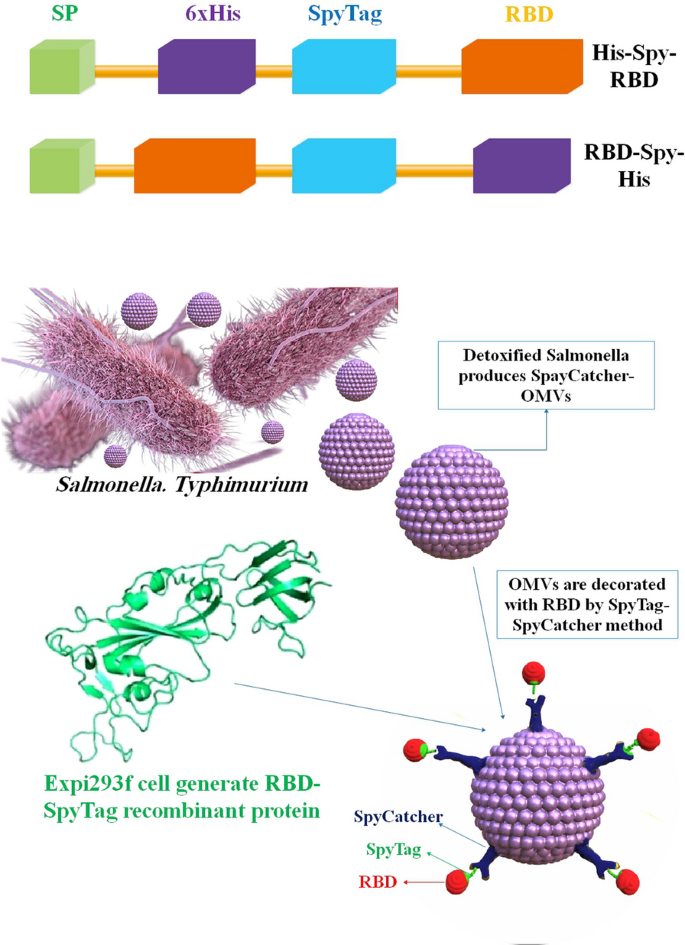
Outline of the building blocks of expressiveness and the adornment of the OMV. Recombinant antigens based on RBDs fused to the N and C termini of SpyTag are designed. A simplified diagram showing how RBD-OMVs are made
Vaccines made from Salmonella may be given through various methods, including orally and nasally. Even while the gut microbiome may neutralize bacteria-based vaccinations administered orally, the risk of unwanted side effects from the adjuvant or viral vector is thought to be lower with these vaccines. When given orally, Salmonella strains are said to exhibit controlled delayed attenuation and stimulate long-lasting humoral and cellular protection. Furthermore, the cost to manufacture vaccines based on attenuated Salmonella is minimal. Anti-coronavirus vaccines based on Salmonella are still in their early stages of research; therefore, this study might pave the way for future COVID-19 vaccinations [87]. Another research project looked at whether modified S proteins from SARS-CoV-2 may be used to treat or prevent COVID-19. It has been established that administering these viral proteins using Salmonella as a transport platform causes the body to produce antibodies against SARS-CoV-2 and activates cellular immune responses against COVID-19. Furthermore, it was shown that Salmonella BRD509, when given orally, did not have significant cytotoxic effects in vivo [88]. In addition, these in vitro and in vivo results support the idea that Salmonella strains constitute a reliable platform for the sustained synthesis of antigen proteins because of their efficient type III secretion system. When Salmonella infects mucosal cells, it can deliver antigens via intracellular patriotization. More robust cellular and humoral immune responses are produced when the immunization against this virus strain is administered orally. The findings of this study make it possible to create safer Salmonella strains that can produce and express antigens of various sizes with fewer inflammatory reactions. These strains could be used as platforms for vaccines with low or no toxicity and the potential for effective antibody production when given orally. Consider Salmonella as a safer oral vaccine development platform for emerging infectious illnesses like COVID-19. Vaccines against COVID-19 could benefit from administration through limoncello strains [83] (Fig. 5).
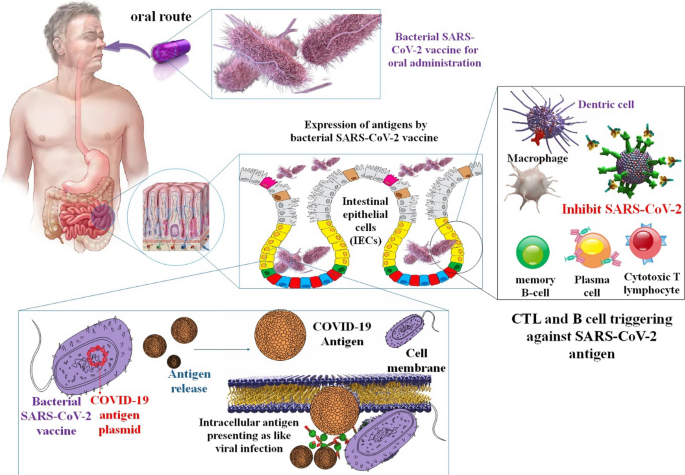
The mechanism by which a bacterial COVID-19 vaccine is administered orally
In an investigation, high-pressure homogenization technology was employed by the researchers to effectively stimulate the bacterial membrane for the production of synthetic vesicles. Additionally, the same technology was utilized to compel the FP cytolysin A (ClyA)-RBD to traverse the apertures in the bacterial membrane, thereby augmenting the interaction between ClyA-RBD and the membrane. By making ClyA look like the support structure of the SARS-CoV-2 S2 protein, researchers could reveal the RBD on the self-assembled bacterial particles very well. High contact, polymerization of ClyA-RBD on bacterial membranes, and the formation of "ring-like" NPs on the vesicles were the results. There was a 107-fold increase in bacterial biomimetic vesicles (BBVs) yield compared to OMV yield, and the RBD was presented on BBVs 28.16-fold more efficiently than on OMVs. Subcutaneously administered RBD-BBVs may localize to lymph nodes, enhance antigen uptake and processing, and stimulate SARS-CoV-2-specific humoral and cellular immune responses in mice [89].
The utilization of bacteria as a delivery system in HIV
HIV, the virus responsible for acquired immunodeficiency syndrome (AIDS), attacks explicitly CD4+ helper T cells and slowly wipes them out. ssRNA molecules are enclosed by a capsid to form the HIV genome [90]. HIV infections start when the C–C chemokine receptor type 5 (CCR5) or C–X–C chemokine receptor type 4 (CXCR4) coreceptor on target cells come into contact with one of the glycoproteins on the surface of the viral capsid, mainly gp120 [91]. To prevent HIV transmission between heterosexual partners, many methods are being researched. Microbicides delivered topically to the vagina are among those that are being actively investigated. Using a topical microbicide to prevent HIV presents several medication delivery difficulties. The vaginal mucosal barriers and probable medication breakdown in the vaginal lumen owing to pH and enzymes present are some of these difficulties. Additionally, new medications being tested as microbicides have unique modes of action that, in some circumstances, need drug targeting to a particular site of action. In HIV prophylaxis, NPs provide a delivery method for targeted or regulated distribution to the vagina [11].
EcN was engineered to produce HIV-gp41-hemolysin. A synthetic peptide that blocks HIV from entering cells by preventing it from joining with them. Because it may persist in the GI and genital tract of mice for weeks to months, engineered EcN shows promise as an anti-HIV microbicide. In an investigation, Mice afflicted with V. cholerae had an improved chance of survival after receiving engineered EcN expressing cholera autoinducer 1. Stable bacterial colonization and intravaginal synthesis of the HIV inhibitor cyanovirin-N were seen after intravaginal delivery of a Lactobacillus strain harboring the gene for cyanovirin-N. Using this method, microorganisms promise as low-cost barriers to HIV transmission [92]. Another study looked at the possibility of employing S. typhi Ty21a BGs as a novel method of delivering an HIV vaccine. DNA vaccines offer great promise for inducing immunological responses since they are stable and straightforward to manipulate. Researchers discovered that murine macrophage RAW264.7 cells effectively produced gp140 after exposure to Ty21a BGs loaded with an HIV gp140 DNA vaccine (Ty21a BG-DNA). Anti-gp120 antibody responses were significantly higher in mice immunized with BGs-DNA than in those immunized with DNA alone. BG-induced IL-10 production was linked to Ab responses via the TLR4 pathway. These results suggest that Ty21a BGs is a unique and efficient DNA vaccine delivery vehicle, offering a new approach to creating HIV vaccines. Despite over three decades of research and development efforts, there is presently no HIV vaccine that can be administered to humans. The Ty21a BGs not only stimulated intestinal mucosal immune responses but also enhanced peripheral antibody responses to the pSV140 DNA vaccination. Biased induction of Th2-type cytokines and TLR4 pathway activation seems to be linked to Ty21a BGs' immune-regulatory effects. Found that HIV-specific IgG and IgA antibodies could be generated in the blood and intestinal mucosa, respectively, using this novel experimental setting [93]. As an additional vector for inducing mucosal immune responses, bacterium-like particles (BLPs) have previously been utilized for several vaccinations against respiratory tract viruses. Mice and guinea pigs were immunized with the vaccine by injecting it into their muscles or giving it to them via their noses. Inducing Env-specific secretory IgA (sIgA) in mucosal locations in mice was shown by administering gp120 trimers bound to BLPs by i.m. injection or i.n. drip. As a result, HIV-1 vaccination using gp120 trimers attached to BLPs shows promise [94].
The utilization of bacteria as a delivery system in HPV
Those who have not yet contracted HPV may benefit from a prophylactic vaccination. That's why it's so important to find ways to treat cancers caused by HPV. Vaccines of the third generation, based on nucleic acids, are a quick and easy way to stimulate adaptive immune responses. For the administration of nucleic acid vaccines, bacteria show great promise as live vectors. LPSs, peptidoglycan, and flagellin are all naturally occurring components of the bacterial vector that are recognized by the immune system and cause it to react strongly [95]. Genetically engineered lactic acid bacteria (LAB) have been shown in several pre-clinical and clinical experiments to successfully produce mucosal, humoral, and cellular immunity in the host (Table 2) [96]. Taken as a whole, the results of these investigations point to a handful of important considerations that might provide direction toward maximizing protein output and enhancing immunological response. The adaptive immune response is improved, and the native immune response is triggered by this introduction. The particular methods by which bacterial vectors make nucleic acids accessible in host cells are not yet entirely known, however, especially for certain species. Some non-pathogenic bacteria, including LAB, and recombinant and attenuated forms of pathogenic bacteria, such as Salmonella, Mycobacterium, Yersinia, Listeria, and Shigella, are thought to be carriers of nucleic acid vaccines. Although there are benefits to using a bacterial delivery method, such as the production of a robust immune response, oral administration, and improved APC targeting, there are also substantial drawbacks that should be taken into account [47]. So far, many mucosal vaccines targeting HPV-16 L1, L2, E2, E6, and E7 antigens have been produced, all based on genetically engineered LAB. Importantly, studies testing the efficacy of these recombinant LAB in eliciting an immune response have reached both phase I and II of clinical trials [96]. Significant results from another research show that Bifidobacterium bifidum, whether given intravenously or orally, successfully promotes antitumor immune responses and reduces tumor development in mice. Intravenous delivery of the probiotic Bifidobacterium bifidum to tumor-bearing mice activates tumor-specific IL-12 and IFN-γ, lymphocyte proliferation, and CD8+ cytolytic responses that suppress and eliminate tumor development, in contrast to oral administration alone. These results indicated that immune system modification by probiotics administered intravenously is an efficient anticancer strategy. Additional research on the immunomodulatory effects of the probiotic Bifidobacterium bifidum in treating cervical cancer is warranted. The current study set out to compare the preventative-therapeutic impact of oral versus intravenous administration of the probiotic Bifidobacterium bifidum on the immune response and tumor growth of C57BL/6 mice harboring transplanted TC-1 cells of HPV-related tumor, expressing HPV-16 E6/E7 oncogenes [97]. Preclinical and clinical investigations of the LAB-based HPV vaccine have led researchers to conclude that LAB is the most promising vehicle for achieving mucosal vaccination goals. The most popular methods for mucosal administration of antigens are the i.n., intravaginal, and oral routes, with the oral route being the most exciting approach for stimulating mucosal immunity by mucosal vaccination. Studies have shown that the most efficient way to induce a mucosal, humoral, and cellular immune response is by oral administration of HPV-16 oncoproteins produced by recombinant L. lactis or L. casei to the gut mucosa [98] (Table 1) (Fig. 6).
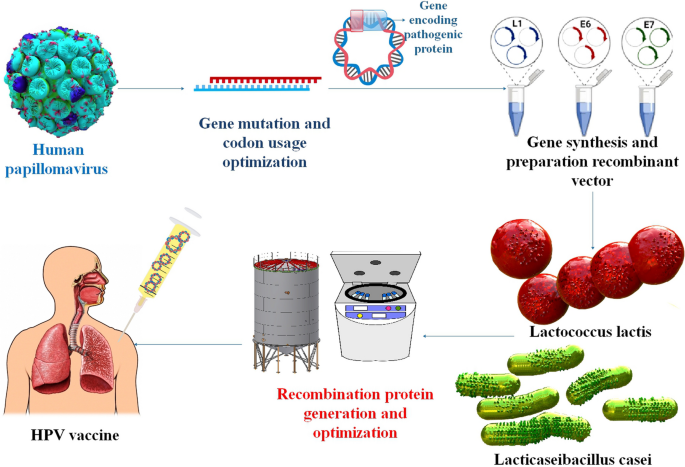
LAB is a typical approach for developing HPV vaccines, and pre-clinical and clinical trial studies have been utilized to determine vaccination effectiveness and safety in mouse models and human test participants
The utilization of bacteria as a delivery system in hepatitis viruses
Viral hepatitis continues to pose a significant threat to human health, as it remains a leading cause of mortality on a global scale [108, 109]. Vaccination is the most reliable method of protection against infectious illnesses, and in some instances, it is the only option. Eliminating deadly diseases like smallpox and polio attests to the efficacy of vaccinations in the past. Human liver-infecting viruses are called hepatitis viruses, and there are five main varieties, designated by the letters A through E [110]. Even though hepatitis A virus (HAV) infection is known to self-resolve with rest and symptomatic therapy, there were 7134 HAV-related fatalities globally in 2016. HBV and the hepatitis C virus (HCV) are expected to have caused 820,000 and 290,000 deaths in 2019, respectively. A satellite virus called hepatitis delta virus (HDV) relies on HBV to produce infectious particles to propagate. The most severe chronic viral hepatitis is thought to result from the co-infection of HDV and HBV. Another oral virus that is often seen in low- and middle-income nations is HEV. It contributed to 44,000 fatalities globally in 2015. Hepatitis A and B vaccinations are currently accessible and safe [111].
Around 71 million individuals worldwide are thought to have chronic HCV infection. Investigations demonstrated that there is currently no licensed vaccination against HCV. A specifically synthesized recombinant FP with a shortened core and HCV NS3 (rC/N) has been expected to be stable and immunogenic. Recombinant proteins are harmless antigens, but without adjuvants, they do not make effective vaccinations [113]. In the recent investigation, BALB/c mice were used to assess the immunogenicity of rC/N as a bipartite antigen combined with N. meningitidis serogroup B OMVs (NMB OMVs). Compared to MF59 and Freund's CIA, NMB OMVs significantly improved Th1 immune responses. A potential method for creating a therapeutic HCV vaccination is rC/N-NMB OMVs. Recombinant protein vaccines are safe and effective, although they often only provide mediocre immunity. Therefore, one or more immune-stimulatory components must be included in the vaccine formulation to stimulate immunological responses that are strong, focused, and persistent in response to the delivered antigen. Microbially derived adjuvants stand out among those legally allowed to be used. The OMVs, which are primarily generated from Gram-negative bacteria, are inherently non-replicating and highly immunogenic. Vesicles produced by detergents may produce various pro-inflammatory cytokines and are safe to use. N. meningitidis serogroup B OMVs (NMB OMVs) have shown that they may be used as adjuvants in various bacterial and viral vaccines to enhance cellular and humoral immune responses. For cancer immunotherapy in mice, the OMVs of E. coli are also employed as an adjuvant and delivery mechanism. To assess the likely immune responses to HCV in a mouse model, this work administered the NMB OMVs simultaneously with the shortened recombinant core1-118 (rCore) and NS31095-1384 (rNS3) FP (rC/N) [114].
HBV virus is an enveloped virus with DNA-based genetic information encapsulated within an icosahedral capsid [112]. The HBcAg-149 was used as a model antigen in a different study to assess and compare the immunogenic capacity of recombinant ghosts employing inner and OM anchor systems. Delivery system particle production may facilitate antigen absorption by APCs, particularly dendritic cells, at the site of infection. These cells subsequently move to lymph nodes where the antigen is presented to naïve T-cells. Empty bacterial cell ENVs (ghosts) were studied for their potential to transport exogenous antigens via two membrane compartments of E. coli. Mice were immunized subcutaneously with BGs containing the HBV core 149 protein (HBcAg-149) as a model antigen, and their immune responses were compared to those of BGs containing a control antigen anchored in the inner membrane of E. coli. Mice exposed to either system developed robust immune responses against the HBcAg-149 target antigen. The findings demonstrate that BGs function splendidly as a vehicle for transporting antigens. By inserting an E-specific transmembrane tunnel structure into their cell wall, Gram-negative bacteria produce BGs when the cloned lysis gene E of bacteriophage PhiX174 is expressed [37].
Several attenuated strains of S. Typhimurium and S. Dublin were found to stably produce hybrid HBV nucleocapsid-pre-S(2) FPs in the presence of aromatic compounds. When these live recombinant bacteria were injected intraperitoneally (i.p.) into BALB/c mice, they produced antibodies against the HBV core antigen (HBc) and the pre-S2 protein. Following oral administration of recombinant salmonellae, the seroconversion rate to anti-HBc in mice differed by salmonella strain. High titer anti-HBc Abs and lower titer anti-pre-S2 serum were seen in the best carrier strain two weeks after a single oral vaccination. The distribution of Ig class and IgG subclass anti-HBc Abs after intramuscular and oral vaccination is supportive of the induction of functional T cell assistance [115].
The utilization of bacteria as a delivery system in influenza virus
A case of the flu, despite seasonal flu vaccination, a virus experiences genetic drift and change, making the general population vulnerable to newly developing pandemic strains. Recombinant OMVs (rOMVs), generated from E. coli, have been suggested as a potentially helpful vaccination strategy that directly connects adjuvant and antigen. OMVs (diameter: 50–200 nm) displaying the target antigen are shed when hyper vesiculating strains of E. coli are transformed with a plasmid containing the transmembrane protein ClyA followed by the target antigen [116]. The rOMVs can be harvested, suspended in a buffer, and employed as a vaccine, obviating additional protein purification or the inclusion of supplementary adjuvants, which is a prerequisite for other expeditious vaccine platforms. The research team has demonstrated that rOMVs containing a heterospecies tandem sequence of peptides derived from the matrix 2 protein ectodomain of influenza (M2e4xHet), comprising of human, swine, and avian species, have the potential to safeguard against various influenza A subtypes in mice with distinct genetic backgrounds. This finding establishes M2e4xHet rOMVs as a promising vaccine candidate for protecting against pandemic influenza A [117]. The study conducted by the researchers demonstrated that the regulated discharge of rOMV constructs exhibits promise as a unipotent vaccine to safeguard against the influenza A challenge. The vaccine generates antibody titers expeditiously, which endure for at least 6 months in mice, thereby providing protection. The utilization of PLGA µP containing M2e4xHet rOMVs has been observed to yield substantial and enduring immunity against the influenza A/PR8 challenge. Prior research involving PLGA µP has involved encapsulating inactivated influenza virus, influenza antigens, and influenza DNA for vaccine development against influenza [116]. The activation of TLR signaling through transported pathogen-associated molecular patterns (PAMPs) occurs upon the uptake of OMV by host cells. Alveolar macrophages are a crucial type of immune cells situated at the interface between air and tissue. They play a pivotal role in defending against inhaled microorganisms and particles, serving as the primary line of defense. The present investigation examined the reaction of primary human macrophages to bacterial vesicles, namely Legionella pneumophila, Klebsiella pneumoniae, E. coli, S. enterica, and Streptococcus pneumoniae. The researchers observed that there was a similar level of NF-κB activation across all the vesicles that were tested. In contrast, scholars depict a distinctive IFN-I signaling pattern characterized by extended STAT1 phosphorylation and robust Mx1 induction, which impedes the replication of influenza A virus solely for Klebsiella, E. coli, and Salmonella OMVs. The antiviral effects induced by OMVs were observed to be comparatively weaker for Clear coli OMVs that were devoid of endotoxin and OMVs that were treated with Polymyxin. The antiviral status was not replicated by LPS stimulation; however, it was nullified by the absence of TRIF due to knockout. Significantly, the supernatant obtained from macrophages treated with OMV elicited an antiviral reaction in alveolar epithelial cells (AEC), indicating the occurrence of intercellular communication induced by OMVs. Ultimately, the findings were verified through an ex vivo infection model utilizing primary human lung tissue. To summarize, the OMVs of Klebsiella, E. coli, and Salmonella prompt antiviral immunity in macrophages through TLR4-TRIF signaling, thereby diminishing viral replication in macrophages, AECs, and lung tissue. Gram-negative bacteria have been observed to stimulate antiviral immunity in the lungs via OMVs. This finding has significant implications for the outcome of bacterial and viral coinfections [118].
Others have also proven that LAB, depending on the strain, may alter respiratory immunity when taken orally. Using a fatal model of influenza virus pneumonia, the ability of many L. plantarum strains to modify respiratory immunity when orally delivered was assessed. The L. plantarum 06CC2 strain was the most effective in increasing the survival rate of influenza-infected mice, whereas the L. plantarum 05AM23, 06TCa8, 06TCa40, and 06CC9 strains had no impact. The administration of the 06CC2 strain through oral means resulted in a decrease in influenza virus titers in the lungs. Additionally, it was observed that there was an improvement in the Th1 response in the respiratory tract, as well as an increase in NK-cell activity in both the lungs and spleens. Furthermore, a comprehensive screening of immunomodulatory LAB was conducted, wherein multiple strains of Lactiplantibacillus were assessed for their capacity to modulate immunity in TNF-α-activated HT-29 cells and peripheral blood mononuclear cells. Of the strains that were evaluated, L. plantarum CNRZ1997 exhibited a noteworthy ability to augment the production of cytokines associated with inflammation in both epithelial and immune cells. Conversely, other strains of L. plantarum either had no impact or displayed anti-inflammatory properties. The study revealed that the CNRZ1997 strain, when administered orally, could diminish the proliferation of the influenza virus in the respiratory tract of mice. The efficacy of oral administration of L. plantarum CNRZ1997 was comparable to that of L. rhamnosus GG or L. casei DN114-001, two commercially available probiotic strains that have demonstrated anti-influenza virus properties in preclinical and human trials, in safeguarding mice against influenza infection [119,120,121,122].
The utilization of bacteria as a delivery system in another virus
Ebola virus (EV), belonging to the Filoviridae family, is a zoonotic pathogen responsible for causing Ebola virus disease (EVD), also called Ebola hemorrhagic fever. The disease is endemic to West Africa and Middle Africa and has resulted in over 34,000 human cases and 15,000 fatalities. Viruses from Zaire (EBOV), Bundibugyo (BDBV), Sudan (SUDV), Tai Forest (TAFV), Reston (RESTV), and Bombali (BOMV) are only some of the six antigenically different species that make up the EV genus. A substantial health danger is posed by EBOV, SUDV, and BDBV because of the high mortality rates associated with their associated diseases in humans (up to 90% in some instances) [123]. In West Africa and Middle Africa, SUDV and EBOV cause serious sickness and, on occasion, death. The BLPs vaccines can potentially provide immunized people with safer and more effective protection against infections. Subunit vaccines have extensively used BLPs, a unique surface display technology for proteins. Nonliving particles generated from gram-positive L. lactis used in food production that have not been genetically changed are the basis of the BLPs surface display technology, which also uses a protein anchor (PA). L. lactis is well suited for use as a vaccine since it has a history of being considered safe by the FDA. AcmA is an autolysin from L. lactis, and its C-terminal peptidoglycan-binding domain provides three lysin motifs (LysM) for the PA [124]. This research demonstrates the immunogenicity in mice of a bivalent BLPs-based vaccination created by combining SUDV-BLPs with EBOV-BLPs at a 1: 1 ratio, called SUDV-EBOV BLPs (S/ZBLP + 2 + P). The SUDV-EBOV BLPs generated both Th1 and Th2 immune responses and caused strong protection against SUDV and EBOV. The findings suggested that a vaccine based on SUDV and EBOV BLPs might be a strong contender in the fight against SUDV and EBOV infections and provide a method for creating universal vaccinations against EVD [123].
The predominant etiological agent of non-bacterial gastroenteritis on a global scale is the human norovirus. In developing nations, it is the second most common cause of diarrheal fatalities in children. Empirical data indicates that noroviruses can adhere to the exterior of commensal bacteria, and the existence of these bacteria has an impact on both acute and persistent murine norovirus infection by influencing the host's immune responses. The interactions between norovirus and bacteria have been found to induce stress responses in the bacteria and result in an increase in the production of BEVs. The researchers postulated that BEVs may impact murine norovirus infection by modulating the antiviral immune response based on their known capacity to readily traverse intestinal barriers and penetrate the lamina propria to modulate host responses. The current investigation demonstrates the capacity of murine norovirus to adhere to purified bacterial vesicles, thereby enabling the simultaneous introduction of virus and vesicle to target cells. Investigations employed S. Typhimurium to investigate the impact of OMVs originating from an intestinal pathogen in contrast to those generated by commensal bacteria. In addition, scholarly investigations have demonstrated that the co-inoculation of macrophages with murine noroviruses and vesicles results in a decrease in viral infection as compared to the infection caused by the virus alone. Co-inoculation with bacterial vesicles leads to increased production and release of pro-inflammatory cytokines in response to viral infection. The findings suggest that bacterial vesicles could potentially function as a means of regulating and curtailing murine norovirus infection, as evidenced by the increased bacterial vesicle production in vivo following infection. This may ultimately serve to restrict the duration of the disease [125] (Table 2).
Comments
Post a Comment